Overview
The quaternary ammonium compounds are widely used as disinfectants. Health-care–associated infections have been reported from contaminated quaternary ammonium compounds used to disinfect patient-care supplies or equipment, such as cystoscopes or cardiac catheters. The quaternaries are good cleaning agents, but high water hardness and materials such as cotton and gauze pads can make them less microbicidal because of insoluble precipitates or cotton and gauze pads absorb the active ingredients, respectively. One study showed a significant decline (~40%–50% lower at 1 hour) in the concentration of quaternaries released when cotton rags or cellulose-based wipers were used in the open-bucket system, compared with the nonwoven spunlace wipers in the closed-bucket system As with several other disinfectants (e.g., phenolics, iodophors) gram-negative bacteria can survive or grow in them.
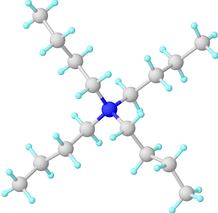
Chemically, the quaternaries are organically substituted ammonium compounds in which the nitrogen atom has a valence of 5, four of the substituent radicals (R1-R4) are alkyl or heterocyclic radicals of a given size or chain length, and the fifth (X-) is a halide, sulfate, or similar radical.
Each compound exhibits its own antimicrobial characteristics, hence the search for one compound with outstanding antimicrobial properties. Some of the chemical names of quaternary ammonium compounds used in healthcare are alkyl dimethyl benzyl ammonium chloride, alkyl didecyl dimethyl ammonium chloride, and dialkyl dimethyl ammonium chloride. The newer quaternary ammonium compounds (i.e., fourth generation), referred to as twin-chain or dialkyl quaternaries (e.g. didecyl dimethyl ammonium bromide and dioctyl dimethyl ammonium bromide), purportedly remain active in hard water and are tolerant of anionic residues.
A few case reports have documented occupational asthma as a result of exposure to benzalkonium chloride.
Mode of Action
The bactericidal action of the quaternaries has been attributed to the inactivation of energy-producing enzymes, denaturation of essential cell proteins, and disruption of the cell membrane. Evidence exists that supports these and other possibilities.
Microbicidal Activity
Results from manufacturers’ data sheets and from published scientific literature indicate that the quaternaries sold as hospital disinfectants are generally fungicidal, bactericidal, and virucidal against lipophilic (enveloped) viruses; they are not sporicidal and generally not tuberculocidal or virucidal against hydrophilic (nonenveloped) viruses. The poor mycobactericidal activities of quaternary ammonium compounds have been demonstrated. Quaternary ammonium compounds (as well as 70% isopropyl alcohol, phenolic, and a chlorine-containing wipe [80 ppm]) effectively (>95%) remove and/or inactivate contaminants (i.e., multidrug-resistant S. aureus, vancomycin-resistant Entercoccus, P. aeruginosa) from computer keyboards with a 5-second application time. No functional damage or cosmetic changes occurred to the computer keyboards after 300 applications of the disinfectants.
Attempts to reproduce the manufacturers’ bactericidal and tuberculocidal claims using the AOAC tests with a limited number of quaternary ammonium compounds occasionally have failed. However, test results have varied extensively among laboratories testing identical products.
Uses
The quaternaries commonly are used in ordinary environmental sanitation of noncritical surfaces, such as floors, furniture, and walls. EPA-registered quaternary ammonium compounds are appropriate to use for disinfecting medical equipment that contacts intact skin (e.g., blood pressure cuffs).
Sunday, June 27, 2021
Refrences
Please Check out file at the following link